What is an Electron?
I already went over electrons in The Atom section, but an electron is a negatively Charged particle, located in the electron cloud of an atom.
Electron Configuration
Electron configurations show how electrons are distributed in an atom.
There are principal energy levels in electron configurations, they are shown as n=1, 2, 3, 4, etc.
The principal energy levels describe the average distance between an electron and nucleus. The energy levels increase the farther away from the nucleus the electrons are. Electrons also occupy sublevels inside each energy level.
I already went over electrons in The Atom section, but an electron is a negatively Charged particle, located in the electron cloud of an atom.
Electron Configuration
Electron configurations show how electrons are distributed in an atom.
There are principal energy levels in electron configurations, they are shown as n=1, 2, 3, 4, etc.
The principal energy levels describe the average distance between an electron and nucleus. The energy levels increase the farther away from the nucleus the electrons are. Electrons also occupy sublevels inside each energy level.
Sublevels
Sublevels match almost exactly to the different areas electrons can be found in an atom. Each sublevel is represented by a letter.
Ex. s, p. d, f, (g)
There are different shapes for each sublevel too.
Sublevels match almost exactly to the different areas electrons can be found in an atom. Each sublevel is represented by a letter.
Ex. s, p. d, f, (g)
There are different shapes for each sublevel too.
|
s Sublevel
"Spherical" |
p Sublevel
"Dumbbell" |
d Sublevel
"Clover" |
f Sublevel
"Double Clover" |
Types of Sublevels in Energy Levels
Atomic Orbitals
For atomic orbitals, every sublevel is positioned differently in 3D space, and this is called Atomic Orbital. Orbitals in a specific sublevel are different in position, but very similar in size. However, they can only hold two electrons.
Number of Atomic Orbitals in Sublevels
For atomic orbitals, every sublevel is positioned differently in 3D space, and this is called Atomic Orbital. Orbitals in a specific sublevel are different in position, but very similar in size. However, they can only hold two electrons.
Number of Atomic Orbitals in Sublevels
Numbers of Electrons in Each Energy Level
Electrons actually like to be in the orbitals the require the least amount of energy. Just think of it like would you rather be running? Or relaxing.
Usually s>p>d>f in energy within each sublevel.
Usually s>p>d>f in energy within each sublevel.
Writing Electron Configurations
Writing electron configurations at first may seem a bit confusing, but once you get it down it's really quite simple. First you need to know how to setup your periodic table.
Writing electron configurations at first may seem a bit confusing, but once you get it down it's really quite simple. First you need to know how to setup your periodic table.
 www.catalog.flatworldknowlege.com
www.catalog.flatworldknowlege.com
For this periodic table all of the purple blocks are in the s sublevel, green are in the p sublevel, red are in the d sublevel, and blue are in the f sublevel.
To find the electron configuration, for let's say Carbon, you first have to start at Hydrogen which is the very top left element. (It might help if you also have another periodic table to look at that has the element symbols on it)
H's electron configuration is 1s^1 because it is on the top row and has 1 electron. Then you move straight across to He and you get 1s^2. You go down one row and start on 2s because it is the second row, but still in the s sublevel. There are two electrons so you have 1s^2, 2s^2, then move straight across. This is where the p's start. Carbon is the second element in the p sublevel, so you have 2 electrons for Carbon in the p sublevel, and you get 2p^2.
Now you put it all together and C's electron configuration is 1s^2, 2s^2, 2p^2.
To find how many electrons C has, you just have to add together all the superscripts. So 2+2+2= 6.Carbon has 6 electrons.
To find the electron configuration, for let's say Carbon, you first have to start at Hydrogen which is the very top left element. (It might help if you also have another periodic table to look at that has the element symbols on it)
H's electron configuration is 1s^1 because it is on the top row and has 1 electron. Then you move straight across to He and you get 1s^2. You go down one row and start on 2s because it is the second row, but still in the s sublevel. There are two electrons so you have 1s^2, 2s^2, then move straight across. This is where the p's start. Carbon is the second element in the p sublevel, so you have 2 electrons for Carbon in the p sublevel, and you get 2p^2.
Now you put it all together and C's electron configuration is 1s^2, 2s^2, 2p^2.
To find how many electrons C has, you just have to add together all the superscripts. So 2+2+2= 6.Carbon has 6 electrons.
Orbital Notation
 www.jahschem.wikispaces.com
www.jahschem.wikispaces.com
Orbital Notation is basically just another way to write the electron configuration. Except with this way you use either lines or circles, in this case lines, and arrows.
Each arrow stands for 1 electron and each line (or circle) can only hold two arrows. You also can't have two arrows facing the same direction on the same line. Since the s sublevel can only hold two total arrows you only need one line (or circle) for each s sublevel.
If you look above at the orbital notation of carbon, you'll see that 1s and 2s each have one line, but 2p has three. Now, you may be wondering how you can tell how many lines you need. Well, like I said, each line can only hold 2 electrons, so since the p sublevel contains 6 electrons, 6 divided by 2 is 3, so you need three lines.
When you have more than one line (or circle), you have to distribute all the arrows one at a time to each line before you can start adding the second arrow to a line.
Each arrow stands for 1 electron and each line (or circle) can only hold two arrows. You also can't have two arrows facing the same direction on the same line. Since the s sublevel can only hold two total arrows you only need one line (or circle) for each s sublevel.
If you look above at the orbital notation of carbon, you'll see that 1s and 2s each have one line, but 2p has three. Now, you may be wondering how you can tell how many lines you need. Well, like I said, each line can only hold 2 electrons, so since the p sublevel contains 6 electrons, 6 divided by 2 is 3, so you need three lines.
When you have more than one line (or circle), you have to distribute all the arrows one at a time to each line before you can start adding the second arrow to a line.
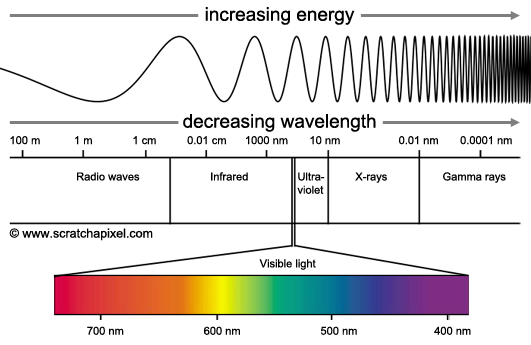
What is the Electromagnetic Spectrum?
The best way to think of the electromagnetic spectrum is to think of it like a piano. You have a bunch of waves arranged like piano keys to make a scale. You have the longer waves on the left, just like you have the lower octave keys on the left of a piano; and the higher frequency more shorter waves on the right of he spectrum, just like you have the higher octave keys on the right of a piano.
The best way to think of the electromagnetic spectrum is to think of it like a piano. You have a bunch of waves arranged like piano keys to make a scale. You have the longer waves on the left, just like you have the lower octave keys on the left of a piano; and the higher frequency more shorter waves on the right of he spectrum, just like you have the higher octave keys on the right of a piano.

This shows the order in which different kinds of rays lie on the electromagnetic spectrum. Radio waves have the lowest frequency, while gamma rays have the highest.
An easy way to remember the order of the rays on the spectrum is:
Rabbits Radio waves
Mate Microwaves
In Infa-red
Very Visible light
Unusual Ultra-violet
eXpensive X-rays
Gardens Gamma rays
(shoutout to home.clara.net for this awesome acronym and fabulous relation between waves and pianos!)
An easy way to remember the order of the rays on the spectrum is:
Rabbits Radio waves
Mate Microwaves
In Infa-red
Very Visible light
Unusual Ultra-violet
eXpensive X-rays
Gardens Gamma rays
(shoutout to home.clara.net for this awesome acronym and fabulous relation between waves and pianos!)
Properties of Waves
 www.misswise.weebly.com
www.misswise.weebly.com
There are many different properties of a wave, the node, the amplitude, the wavelength, the trough, and the peak, which is also referred to as the crest.
Wave- A vibrating disturbance where energy is transmitted
Node- Point of zero amplitude
Amplitude- Height of the wave from midline to either the trough or crest of the wave
Wavelength- The distance from two points that are identical on a wave.
Ex. From trough to trough
Trough- Lowest point on a wave
Crest- Highest point on a wave
Wave- A vibrating disturbance where energy is transmitted
Node- Point of zero amplitude
Amplitude- Height of the wave from midline to either the trough or crest of the wave
Wavelength- The distance from two points that are identical on a wave.
Ex. From trough to trough
Trough- Lowest point on a wave
Crest- Highest point on a wave
Electromagnetic Radiation
Light is Electromagnetic Radiation, which is just radiation that is made up from electric and magnetic fields.
Speed of Waves
EMR travels at the speed of light (c). The speed is related to the wavelength and frequency of waves.
c= λν
c- speed of light (2.998x10^8 m/s)
λ- wavelength (m or mm)
v- frequency (s^-1 or Hz)
Wavelength and frequency are indirectly related
A scientist named Max Planck discovered that molecules and atoms give off discrete quantities of energy called Quanta.
Quanta- small burst of energy
The formula used to describe this relationship is:
E= hv
E- energy of a vibrating system (J)
h- Planck's Constant (6.626x10^-34 Js)
v- frequency (s^-1 or Hz)
Energy and frequency are directly related
Light is Electromagnetic Radiation, which is just radiation that is made up from electric and magnetic fields.
Speed of Waves
EMR travels at the speed of light (c). The speed is related to the wavelength and frequency of waves.
c= λν
c- speed of light (2.998x10^8 m/s)
λ- wavelength (m or mm)
v- frequency (s^-1 or Hz)
Wavelength and frequency are indirectly related
A scientist named Max Planck discovered that molecules and atoms give off discrete quantities of energy called Quanta.
Quanta- small burst of energy
The formula used to describe this relationship is:
E= hv
E- energy of a vibrating system (J)
h- Planck's Constant (6.626x10^-34 Js)
v- frequency (s^-1 or Hz)
Energy and frequency are directly related
Photoelectric Effect
In 1905, Albert Einstein advanced Planck's theory by studying the photoelectric effect. The photoelectric effect is basically just saying that light contains tiny little "lumps" of light called photons.
The difference between photons and waves is that a photon is a tiny particle of light while a wave is just a stream of photons. This is also called the dual nature of light.
A cool thing about electrons is that they absorb energy, which makes them move to a higher energy level, but when they lose that energy they emit a photon. This photon matches up to a certain frequency of light in the visible spectrum.
In 1905, Albert Einstein advanced Planck's theory by studying the photoelectric effect. The photoelectric effect is basically just saying that light contains tiny little "lumps" of light called photons.
The difference between photons and waves is that a photon is a tiny particle of light while a wave is just a stream of photons. This is also called the dual nature of light.
A cool thing about electrons is that they absorb energy, which makes them move to a higher energy level, but when they lose that energy they emit a photon. This photon matches up to a certain frequency of light in the visible spectrum.
Quantum Mechanical Model
The quantum mechanical model of the atom went through many trial and errors before Erwin Schroedinger finally took into consideration the particle and wave-like nature of an electron and developed equations that did the same. These equations describe the energy of an electron exactly, but there is still a large uncertainty in where the electron is located.
The solutions to these equations are called wave functions. A wave function is an orbital.
Quantum numbers are used to solve these equations. Each quantum number has a certain value for exactly that reason.
However, Schroedinger's equation only solved exactly for Hydrogen, but chemist's and physicist's get around this by making approximations and assumptions that the difference isn't that great.
We still use Schroedinger's model today!
The quantum mechanical model of the atom went through many trial and errors before Erwin Schroedinger finally took into consideration the particle and wave-like nature of an electron and developed equations that did the same. These equations describe the energy of an electron exactly, but there is still a large uncertainty in where the electron is located.
The solutions to these equations are called wave functions. A wave function is an orbital.
Quantum numbers are used to solve these equations. Each quantum number has a certain value for exactly that reason.
However, Schroedinger's equation only solved exactly for Hydrogen, but chemist's and physicist's get around this by making approximations and assumptions that the difference isn't that great.
We still use Schroedinger's model today!