Lewis Dot Structures
These are sometimes called Electron Dot Structures, and all they really are is a way to show how many valence electrons are in an atom.
Valence Electron- Electrons in the highest occupied energy level of an atom
These are sometimes called Electron Dot Structures, and all they really are is a way to show how many valence electrons are in an atom.
Valence Electron- Electrons in the highest occupied energy level of an atom

Valence Electrons must be labeled like this on a Lewis Dot Structure:
Where X is the element, and the numbers correspond to the valence electrons.
Lewis Dot Structures have to follow the Octet Rule, and the octet rule says that atoms like to have full outer shells containing only eight electrons.
Octet- A set of eight
Where X is the element, and the numbers correspond to the valence electrons.
Lewis Dot Structures have to follow the Octet Rule, and the octet rule says that atoms like to have full outer shells containing only eight electrons.
Octet- A set of eight
A shortcut to finding how many valence electrons an element has, is to look at what group it's in.
Group 1 has 1 valence electron
Group 2 has 2
Group 3-12 have 2
Group 13 has 3
Group 14 has 4
Group 15 has 5
Group 16 has 6
Group 17 has 7
Group 18 has 8
Group 1 has 1 valence electron
Group 2 has 2
Group 3-12 have 2
Group 13 has 3
Group 14 has 4
Group 15 has 5
Group 16 has 6
Group 17 has 7
Group 18 has 8
Lewis Dot Structures
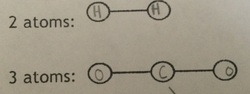
There are different structures for different dot diagrams, but how they look really just depends on how many valence electrons are being shared, and how many elements you are using.
.= 1 electron -------- = 2 electrons
There are different structures for different dot diagrams, but how they look really just depends on how many valence electrons are being shared, and how many elements you are using.
.= 1 electron -------- = 2 electrons
|
|
|
Polarity
In polarity there are two types of bonds, polar and nonpolar.
In polarity there are two types of bonds, polar and nonpolar.
|
Polar bonds
Bonds where the electrons are shared unequally between atoms. The atom that is more electronegative will pull the electrons closer to itself If the atoms are different, they will each have a different pull on the electrons. Ex. HF Fluorine is more electronegative so the shared electrons are held closer to F. |
Nonpolar Bonds
Electrons are shared equally between the two atoms Atoms that are the same, have the same pull on the electrons (they have the same electronegativity value) Ex. H2 H--H They both have the same EN value CCl4 |
All The Cl's should have a "d-" next to it. This means that it is more electronegative than Carbon is.
S- -Higher desity around bonding electrons of the more EN atom (More Electronegative)
S+ -Density is taken away giving it a partial positive charge. (Less Electronegative)
The arrows point in the direction of the more electronegative.
S- -Higher desity around bonding electrons of the more EN atom (More Electronegative)
S+ -Density is taken away giving it a partial positive charge. (Less Electronegative)
The arrows point in the direction of the more electronegative.
How to Tell if it's Polar or Nonpolar
If the atoms around the central atom are different, it will always be polar.
If the atoms around the central atom are the same, it will always be nonpolar.
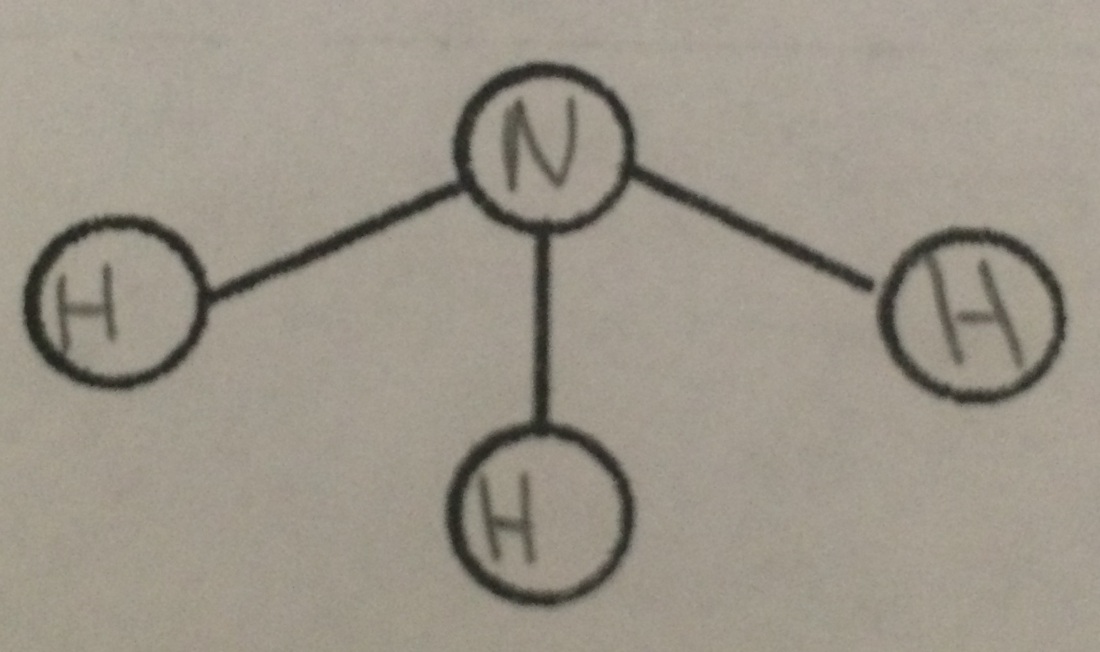
Bent and Pyramid will always be polar.
If the atoms around the central atom are different, it will always be polar.
If the atoms around the central atom are the same, it will always be nonpolar.
Bent and Pyramid will always be polar.
Molecular Geometry
Molecular Geometry is just the shape of molecules. The VSEPR Theory, or Valence Shell Electron Pair Repulsion Theory, states that the electron pairs (both shared and unshared) in the outermost energy level try to get as far away from each other as possible. This is what shapes the molecules.
Molecular Geometry is just the shape of molecules. The VSEPR Theory, or Valence Shell Electron Pair Repulsion Theory, states that the electron pairs (both shared and unshared) in the outermost energy level try to get as far away from each other as possible. This is what shapes the molecules.