International System of Units:
If you're trying to get everyone to understand what your measurements mean, then you have to agree on the units that are going to be used. The International System of Units has been used for all scientific work. It was adopted in 1960, by the General Conference on Weights and Measures.
Another name for the International System of Units is SI. There are seven basic units in SI.
If you're trying to get everyone to understand what your measurements mean, then you have to agree on the units that are going to be used. The International System of Units has been used for all scientific work. It was adopted in 1960, by the General Conference on Weights and Measures.
Another name for the International System of Units is SI. There are seven basic units in SI.
Image made available by www.du4.org
SI Prefixes
Prefixes can be put in front of the base units. They are used to represent quantities that are either larger or smaller than the base units.
Prefixes can be put in front of the base units. They are used to represent quantities that are either larger or smaller than the base units.
|
Prefix
giga mega kilo hector deca deci centi milli micro nano |
Unit Abbreviation
G M k h da d c m µ n |
Meaning
1x10^9 1x10^6 1x10^3 1x10^2 1x10^1 Base 1 (g, m, l ,sec) 1x10^-1 1x10^-2 1x10^-3 1x10^-6 1x10^-9 |
Unit Conversion
In order to be able to convert metric units, you have to learn how to do a process called "Dimensional Analysis." Dimensional Analysis, or Factor Label Method, is a problem-solving method that uses the fact that any number or expression can be multiplied by one without changing its value. It is a useful technique. The only danger is that you may end up thinking that chemistry is simply a math problem - which it definitely is not. Conversion factors are formed from equalities that state a relationship between two units.
Conversion Factors- numbers that are used to change from one unit to another.
When Conversion factors are used properly, all of the units will divide out, or cancel out, except for the unit being converted.
For example: How many kilometers are equal to 250 meters?
250m| 1 km = 0.250 km
| 1x10^3m
To set up this problem you have to put what you are trying to convert on the outside of the fence (250m). Then on the bottom you put the prefix value. You put the unit that will cancel out what you are starting with on the bottom. in this case it is 1x10^3, because the prefix is "kilo", and on the top you have to put what you are converting to (1 km).You put the 1 with the km because "km" has more letters than "m" so you always put the 1 with whichever has more letters.
To solve you have to multiply 250m by 1km, and then divide by 1x10^3
In order to be able to convert metric units, you have to learn how to do a process called "Dimensional Analysis." Dimensional Analysis, or Factor Label Method, is a problem-solving method that uses the fact that any number or expression can be multiplied by one without changing its value. It is a useful technique. The only danger is that you may end up thinking that chemistry is simply a math problem - which it definitely is not. Conversion factors are formed from equalities that state a relationship between two units.
Conversion Factors- numbers that are used to change from one unit to another.
When Conversion factors are used properly, all of the units will divide out, or cancel out, except for the unit being converted.
For example: How many kilometers are equal to 250 meters?
250m| 1 km = 0.250 km
| 1x10^3m
To set up this problem you have to put what you are trying to convert on the outside of the fence (250m). Then on the bottom you put the prefix value. You put the unit that will cancel out what you are starting with on the bottom. in this case it is 1x10^3, because the prefix is "kilo", and on the top you have to put what you are converting to (1 km).You put the 1 with the km because "km" has more letters than "m" so you always put the 1 with whichever has more letters.
To solve you have to multiply 250m by 1km, and then divide by 1x10^3
Volume and Density
Volume- the space occupied by a sample of matter.
Density- how compact a substance is.
Mass- how much matter an object contains.
The cubic meter (cm^3) is the SI unit for measuring volume. When chemists measure the volumes of liquids and gases they often use a non-SI unit called the Liter (L). Both units, mL and cm^3 are interchangeable.
The kilogram (kg) is the SI base unit for measuring mass.
In order to find the density of something you can use the formula
D = M
V
Where D is density, M is mass, and V is volume.
Volume- the space occupied by a sample of matter.
Density- how compact a substance is.
Mass- how much matter an object contains.
The cubic meter (cm^3) is the SI unit for measuring volume. When chemists measure the volumes of liquids and gases they often use a non-SI unit called the Liter (L). Both units, mL and cm^3 are interchangeable.
The kilogram (kg) is the SI base unit for measuring mass.
In order to find the density of something you can use the formula
D = M
V
Where D is density, M is mass, and V is volume.
Water Displacement Method
The water displacement method is a way to determine an object's volume.When you put an object into water it will displace (or push out) some of the water. Measuring how the water levels change after putting an object into it is an easy and accurate way measuring the volume of the object.
The water displacement method is a way to determine an object's volume.When you put an object into water it will displace (or push out) some of the water. Measuring how the water levels change after putting an object into it is an easy and accurate way measuring the volume of the object.
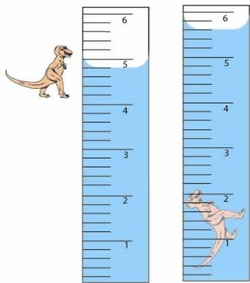
You can see that the container on the left is at 4.8 mL and the container on the right is at 5.6 mL. SO then you subtract $.8 from 5.6 and you get 0.8 mL.
The volume of the toy dinosaur is 0.8 mL
Image made available by www.cstephenmurray.com
The volume of the toy dinosaur is 0.8 mL
Image made available by www.cstephenmurray.com
Scientific Measurements
A measurement is a type of observation. There are two types of observations, Qualitative and Quantitative.
Qualitative Measurements- Descriptive
Ex. hot, cold, heavy, light, big, blue, furry
Quantitative Measurements- Observations made with a measuring instrument and includes both a number and a unit.
Ex. Ruler, balance, thermometer, graduated cylinder
Percent Error
Percentage Error is a way for scientists to find how far off a laboratory value is from the commonly accepted value.
The formula for finding the percentage error is
% error = | Accepted Value - Experimental Value | x100
| Accepted Value |
To get the percent error you take the absolute value of the Accepted Value minus the Experimental value. Divide by Accepted value. Multiply by 100.
A measurement is a type of observation. There are two types of observations, Qualitative and Quantitative.
Qualitative Measurements- Descriptive
Ex. hot, cold, heavy, light, big, blue, furry
Quantitative Measurements- Observations made with a measuring instrument and includes both a number and a unit.
Ex. Ruler, balance, thermometer, graduated cylinder
Percent Error
Percentage Error is a way for scientists to find how far off a laboratory value is from the commonly accepted value.
The formula for finding the percentage error is
% error = | Accepted Value - Experimental Value | x100
| Accepted Value |
To get the percent error you take the absolute value of the Accepted Value minus the Experimental value. Divide by Accepted value. Multiply by 100.
Accuracy vs Precision
Accuracy- How close a measurement is to the true or accepted value.
Ex. Weighing a 50g mass
50.00g- Accurate
32.18g- Not accurate
49.99- Accurate
Precision- How close multiple measurements are to each other.
Ex. Taking data for weighing a 50g mass
Accurate, Precise
50.00g
50.00g
50.00g
Not accurate, Precise
32.18g
32.18g
32.18g
Accurate, Precise
50.00g
49.99g
50.00g
Scientific Notation
Scientific notation is a special way of writing numbers. It makes it easier for scientists to handle very large numbers, and very small numbers.
Ex. 556,000 is 5.56x10^5
0.0000000794 is 7.94x10^-8
If the number is greater than 1, you move the decimal to the left until there is only one Significant Figure in front of it. than you take that number and multiply it by 10 to the however many decimal places you moved, power. If the number is less than 1 you move the decimal to the right until there is only one Sig. Fig. in front of it. Then you take that numbe and multiply it by 10 to the however many decimal places you moved, negative power.
For instance in the first example, you have 556,000. You move the decimal 5 places to the left and get 5.56x10^5
In the second example, you have 0.0000000794. You move the decimal 8 places to the right, and since you moved it to the right it gets a negative. so it ends up being 7.94x10^-8
Scientific notation is a special way of writing numbers. It makes it easier for scientists to handle very large numbers, and very small numbers.
Ex. 556,000 is 5.56x10^5
0.0000000794 is 7.94x10^-8
If the number is greater than 1, you move the decimal to the left until there is only one Significant Figure in front of it. than you take that number and multiply it by 10 to the however many decimal places you moved, power. If the number is less than 1 you move the decimal to the right until there is only one Sig. Fig. in front of it. Then you take that numbe and multiply it by 10 to the however many decimal places you moved, negative power.
For instance in the first example, you have 556,000. You move the decimal 5 places to the left and get 5.56x10^5
In the second example, you have 0.0000000794. You move the decimal 8 places to the right, and since you moved it to the right it gets a negative. so it ends up being 7.94x10^-8
Significant Figures
All measurements have uncertainty. The "certain" digits include all of the umbers that are read off of the device you're using for measurement, plus one extra estimated digit.
All measurements have uncertainty. The "certain" digits include all of the umbers that are read off of the device you're using for measurement, plus one extra estimated digit.
 Image made available by www.quia.com
Image made available by www.quia.com
For Example
This is a graduated cylinder, and it is measured in mL.
The proper reading for the amount of liquid in the cylinder is 15.0 mL.
0 is the estimated value.
This is a graduated cylinder, and it is measured in mL.
The proper reading for the amount of liquid in the cylinder is 15.0 mL.
0 is the estimated value.
The amount of definite digits really depends on what you are measuring with. An exact number doesn't have any uncertainty, giving it an infinite amount of significant figures.
Ex. 17 frogs, 17.00000000000...
However, defined quantities are considered to be exact.
Ex. 12 inches = 1 foot, 1000 milliliters = 1 liter
Rules for Sig Figs
1. All non-zero digits are significant
Ex. 4.37- 3 s.f.
4.8253- 5 s.f.
2. Zeros on the left are never significant (leading zeros)
Ex. 0.0098- 2 s.f.
0.000004- 1 s.f.
3. Zeros in the middle are always significant (captive zeros)
Ex. 308- 3 s.f.
10098- 5 s.f.
4. Zeros at the end are sometimes significant (trailing zeros)
a) They are significant if the number contains a decimal point.
Ex.129.0- 4 s.f.
0.780- 3 s.f.
b) They are not significant if the number does not contain a decimal point.
Ex. 1660- 3 s.f.
8300- 2 s.f.
Ex. 17 frogs, 17.00000000000...
However, defined quantities are considered to be exact.
Ex. 12 inches = 1 foot, 1000 milliliters = 1 liter
Rules for Sig Figs
1. All non-zero digits are significant
Ex. 4.37- 3 s.f.
4.8253- 5 s.f.
2. Zeros on the left are never significant (leading zeros)
Ex. 0.0098- 2 s.f.
0.000004- 1 s.f.
3. Zeros in the middle are always significant (captive zeros)
Ex. 308- 3 s.f.
10098- 5 s.f.
4. Zeros at the end are sometimes significant (trailing zeros)
a) They are significant if the number contains a decimal point.
Ex.129.0- 4 s.f.
0.780- 3 s.f.
b) They are not significant if the number does not contain a decimal point.
Ex. 1660- 3 s.f.
8300- 2 s.f.

